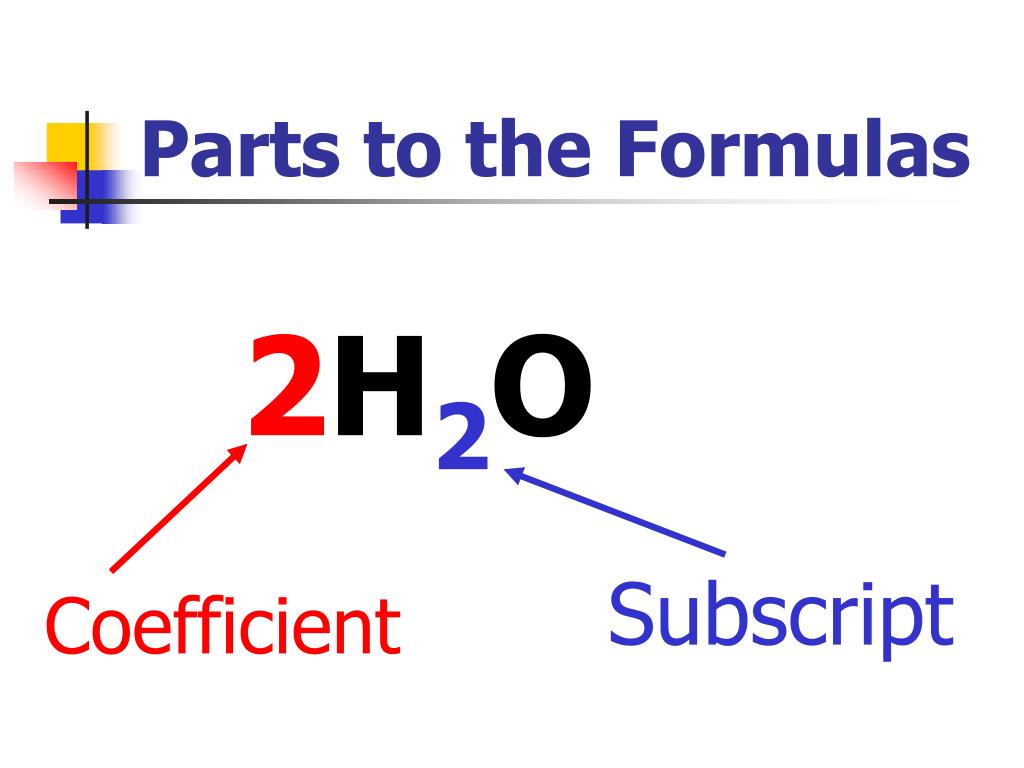
How these names are assigned is beyond the scope of this module. Subscripts indicate the number of atoms or ions in a single unit. There are millions of known molecules that each has its own systematically assigned name. Subscripts are numerals which appear after a chemical symbol in a chemical formula. Some more common molecules and their chemical formulas are:Ĭarbon Dioxide -> CO 2 Ammonia -> NH 3 Glucose/Sugar -> C 6H 12O 6 Isopropyl/Rubbing Alcohol -> C 3H 7OH Table Salt -> NaCl The graphic below illustrates the formula for water using symbols. A subscript is only used when more than one atom is being represented, that is a subscript of "1" will never be seen in a chemical formula. 
The subscript "2" means that two atoms of the element hydrogen have joined together to form a molecule. If we want to represent two atoms of hydrogen, instead of writing H H, we write H 2. For example, "H" represents one atom of hydrogen and "O" represents one atom of oxygen. In chemical formulas the abbreviations of element names, which are listed in the periodic table, are used. Identify the chemical formulas for several common substances.Ĭhemical formulas are used to describe molecules and compounds.Explain how chemical formulas can be used to describe elements and molecules.  
After reading this section you will be able to do the following:
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
